CES 2024 – Valencell Unveils Valencell Health™ and the Fingertip™ Blood Pressure Monitor
 January 9, 2024
January 9, 2024
Valencell Unveils Valencell Health™ and the Fingertip™ Blood Pressure Monitor
The Calibration-Free, Cuffless BPM Solution Targeting Over-the-Counter Use
Pioneers of biometric technology enter the digital health space, developing own-branded solutions for chronic disease management
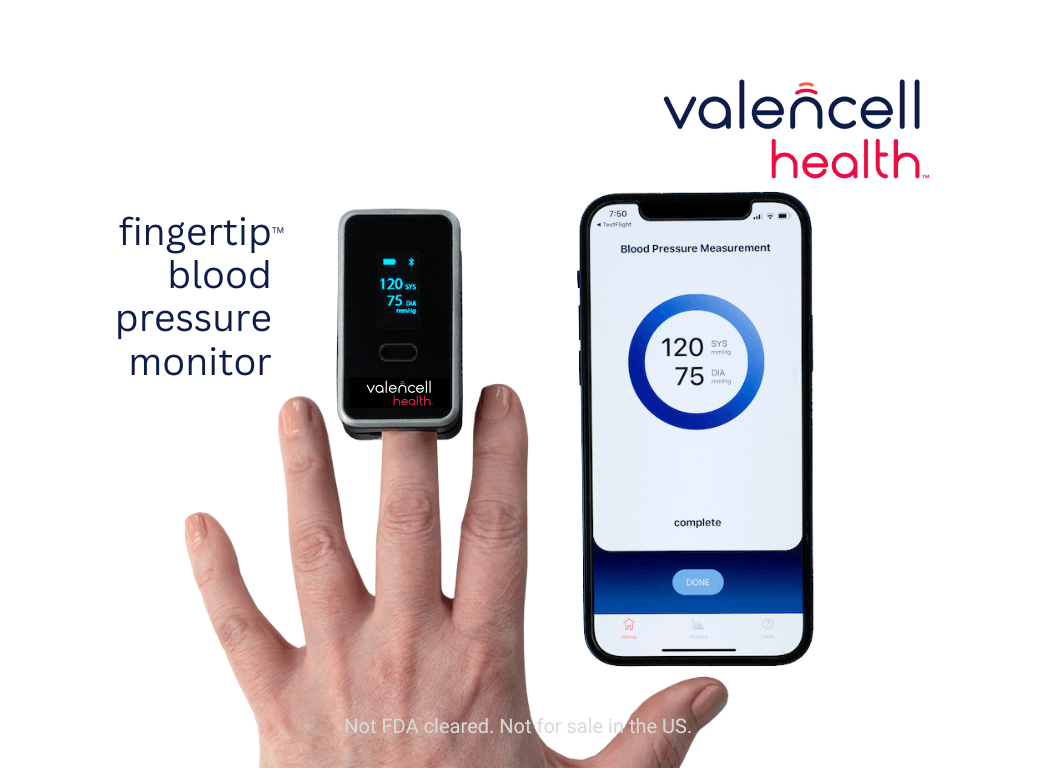
Raleigh, NC & CES Las Vegas, – Valencell, the company whose optical heart rate technology enabled the ability of wearables to accurately measure cardiovascular vitals during exercise, today announced plans to launch its own branded product line in the digital health sector under the name Valencell Health™ as it concentrates efforts to bring solutions to market to manage chronic diseases. The company’s first product candidate is the Fingertip™ Blood Pressure Monitor, focused on helping people spot-check measure BP and manage hypertension by combining an intuitive app with an innovative over-the-counter device to accurately measure blood pressure (BP) from the finger without a cuff or calibration.
Founded in 2006 with a vision to help people live longer, healthier lives, Valencell’s proven inventions, sensor technology, and algorithms can be found in tens of millions of wearable and hearable devices, in over 90 products, for companies such as Samsung, Suunto, Bose, Jabra, Huawei, and Sony. While Valencell was established as a technology provider in consumer sports and fitness wearables, its foundational technology has the potential to realize the dream of seamless chronic disease management through easy-to-use, high-compliance digital health solutions.
The launch of the Valencell Health Fingertip™ BPM device will further the company’s vision of bringing life-changing digital health solutions to consumers and is a strategic evolution for the company. Having developed solutions for some of the world’s leading wearable companies, Valencell is now driven to bring its own portfolio of digital therapeutics to market. Powered by its patented biometric sensor technology, over 150 patents, proprietary AI models, and analytics that have been clinically validated using thousands of datasets over many years, it begins with a focus on hypertension.
“Having spent seventeen years perfecting the ability to capture health data from multiple areas of the body and developing solutions for third parties, I am excited for Valencell to embark on a new chapter. In reviewing our company strategy, we concluded developing our own solutions was the most effective way to get to market faster since the lion’s share of the regulatory and research development already falls upon Valencell,” said Dr. Steven LeBoeuf, President, and co-founder of Valencell. “Beginning with the number one silent killer, hypertension, Valencell is developing digital health solutions that take out all the friction and barriers to capturing and acting on health data to help make it easier for people to take control over their health, live longer, healthier, and more productive lives.”
Meeting Demands Under Pressure
Valencell’s first chronic disease management solution aims to help those with hypertension better and more regularly measure blood pressure and adhere to treatment.
The device has been engineered to measure blood pressure in less than a minute using a small probe on the middle finger without the need for bulky or painful cuffs. Photoplethysmography (PPG) sensors within the device use reflected light to measure blood flow patterns. Proprietary AI algorithms, developed from more than 20,000 PPG datasets comprising more than 10,000 patients, are used to process this information with physical characteristics (age, weight, height and biological sex) to calculate a blood pressure measurement. Diastolic and systolic results are then displayed on the device’s built-in screen and transmitted via Bluetooth® to the app.
Unlike other technologies, the Fingertip™ Blood Pressure Monitor does not need to be calibrated to a BP cuff, a capability that has so far eluded the industry. Valencell’s solution is expected to be the world’s first cuffless and truly calibration-free blood pressure monitor in a fingertip form factor, with the company targeting FDA Clearance in late 2024. Altogether, this has the potential to enable the world’s first over-the-counter cuffless BP monitoring solution.
Although the device has been designed as a stand-alone blood pressure monitor, it has the potential to become a powerful tool to help manage hypertension when paired with its accompanying app. The easy-to-use mobile app has been designed to track and store readings and allow users to set reminders to take their blood pressure. Users will also have the ability to view trends over time, track the frequency of their measurements, and download and share data.
The Heart of The Problem
Globally, more than a billion people suffer from hypertension, and it accounts for approx. ten million deaths per year, yet often hypertension has little or no symptoms and is difficult to manage on an ongoing basis. According to the CDC[1], hypertension affects nearly one-third of Americans, or 75 million people, aged 18 or older. Additionally, for approximately one-half of adults with hypertension (nearly 35 million people), it is uncontrolled, costing $131 billion a year in healthcare costs in the US alone[2].
Critical to managing hypertension and saving lives are regular and reliable BP measurements, yet in a nationwide survey, 62% of Americans with hypertension only measure their BP a few times a month or less, far below expert recommendations of twice daily[3]. However, in a study of consumers that monitored BP at home, 80% of respondents stated they would increase the frequency of measurements using a fingertip device[4].
“The ability to accurately measure blood pressure is essential in ensuring those with cardiovascular conditions adhere to treatments and lifestyle changes their physicians recommend. Anything that makes this easier for people to do can save lives, dollars, and time,” commented Dr. Bill Kraus, Professor of Medicine, Duke University of Medicine. “The rise of digital health solutions and coaching apps are a welcomed trend but are only as good as the data that goes into them. Using the Valencell device that is as simple and painless as pulse oximeters could improve treatment adherence and outcomes and would be a welcomed solution to a largely unmet clinical need.”
Future of Digital Therapeutics
With new algorithms, form factors, and clinical data, blood flow patterns have the potential to measure health statistics ranging from respiratory rates, oxygen levels, cardiac efficiency, and blood pressure. The ability to place these capabilities into ubiquitous, everyday objects that are so intuitive they make it easy for consumers to manage their own health brings the promise of genuinely personal, preventative medicine close to reality.
In the future, Valencell plans to extend its hypertension management offering towards passive blood pressure monitoring in popular wearable form-factors, leveraging its technology. Additionally, the company is investigating how its foundational technology can be applied within these form-factors towards managing diabetes, kidney disease, and coronary artery disease.
To learn more about Valencell visit www.valencell.com. To experience it for yourself during CES, please contact Rebecca Cook by email [email protected] to schedule a demo.
###
About Valencell
Valencell develops and delivers innovative, medical-grade digital health solutions focused on chronic disease management. Our intuitive, accurate solutions make it easier for consumers to manage their own health. Valencell’s patented biometric sensor technology, proprietary algorithms, and analytics, used by third-party wearable solutions on the market today, have been clinically validated in thousands of diverse participants over many years. Valencell is a member of the American Heart Association Center for Health Technology & Innovation who is focused on building and fostering health technology relationships to develop innovative and scalable solutions.
[1] https://pubmed.ncbi.nlm.nih.gov/27855138/
[2] Journal of American Heart Association Trends in Healthcare Expenditures Among US Adults With Hypertension: National Estimates, 2003-2014 – https://pubmed.ncbi.nlm.nih.gov/29848493/
[3] Mayo Clinic – Get the most out of home blood pressure monitoring – https://www.mayoclinic.org/diseases-conditions/high-blood-pressure/in-depth/high-blood-pressure/art-20047889#
[4] Valencell proprietary target audience study completed in October 2022 among 300 consumers ages 21-75 who monitored their own BP at home or at work.